|
Radiation: A Review of Human Carcinogens. International Agency for Research on Cancer.The three share the place in the periodic table assigned to atomic number 1 and hence are called isotopes (from the Greek isos, meaning same, and topos, signifying place) of hydrogen. Atoms are composed of a cloud of electrons surrounding a dense nucleus that is 100,000 times smaller and comprised of protons and neutrons. This means that a neutron in a carbon 14 atom turns into a proton and an. Three nuclei with one proton are known that contain 0, 1, and 2 neutrons, respectively. Internalized radionuclides that emit β particles are carcinogenic to humans (Group 1) ( 1). Carbon 14 decays into nitrogen 14 by beta decay. Ingestion, inhalation, puncture, wound, skin contamination (absorption) Predicted LC-MS/MS Spectrum - 40V, Negative Predicted LC-MS/MS Spectrum - 20V, Negative 
Predicted LC-MS/MS Spectrum - 10V, Negative Predicted LC-MS/MS Spectrum - 40V, Positive Despite this short half-life compared to the age of the earth, carbon-14 is a naturally occurring isotope. Predicted LC-MS/MS Spectrum - 20V, Positive Radioactive carbon-14 has a half-life of 5730 years and undergoes decay, where the neutron is converted into a proton, an electron, and an electron antineutrino: Beta-decay of C-14 nucleus. For example, carbon-14 is a radioactive isotope of carbon that has six protons and eight neutrons in its nucleus. We call forms of elements that have a different number of neutrons, isotopes. 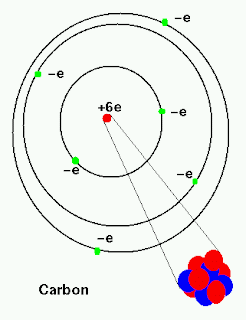
But some types of carbon have more than six neutrons. 
Predicted LC-MS/MS Spectrum - 10V, Positive These are acyclic branched or unbranched hydrocarbons having the general formula CnH2n+2, and therefore consisting entirely of hydrogen atoms and saturated carbon atoms. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. It is one of the isotopes used in radiometric dating of organic material. Element Carbon (C), Group 14, Atomic Number 6, p-block, Mass 12.011. It is has a total of 14 subatomic particles in its nucleus and decays with a half-live of 5,700 years. Belongs to the class of organic compounds known as alkanes. Carbon-14: Carbon-14 is an unstable isotope of carbon.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
